Carbon dioxide (CO2) gas is commonly used in the manufacture of popular beverages. So what is carbon dioxide, why is it dangerous and why do we have to monitor it carefully?
Properties of CO2
Carbon dioxide occurs naturally in our atmosphere at very low concentrations and we produce it when we breathe out. However, it is heavier than air and if it escapes, will tend to sink to the floor, where it can form deadly, invisible pockets. CO2 collects in cellars and at the bottom of containers and confined spaces such as tanks and silos. This is why it is a hazard throughout drinks manufacturing processes, right through to packaging and bottling and even to the bars and eating establishments where the drinks are served.
Why is it dangerous?
Exposure to elevated levels of CO2 can have life threatening effects. As little as 0.5% by volume CO2 can be hazardous to health. Concentrations greater than 10% by volume can kill quite quickly. Because CO2 is completely odourless and colourless, there may well be no indication of danger until it is too late.
Why monitor CO2?
Some still think that monitoring O2 levels protects you from the toxic effects of CO2. This is not the case, however, and here is why.
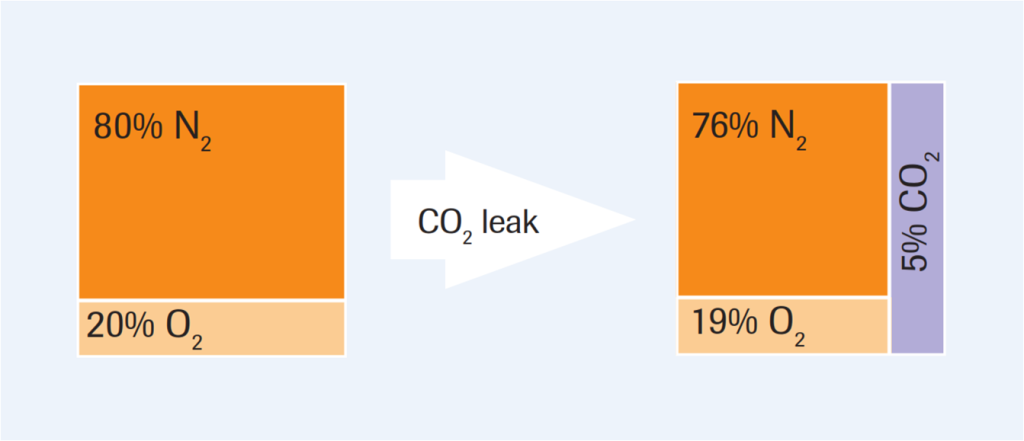
At almost 80% volume, nitrogen (N2) comprises the majority of normal air. Therefore, it is mainly N2 that is displaces by a CO2 leak (fig 1). The percentage increase of CO2 is not matched by a similar decrease in the O2 concentration. Carbon dioxide can exceed safe levels, but O2 levels could still be comparatively unaffected and so considered safe. An additional danger in this case is that, with high concentrations of CO2, some oxygen sensors give a small upscale signal, i.e. a falsely high reading.
Many countries set statutory workplace exposure limits to protect against the effects of a toxic gas. These are generally defined in time weighted averaged. To prove compliance, the CO2 levels each worker is exposed to must be tracked using personal monitors. This should be supported by fixed detectors in at-risk production environments. This is the best way to be sure that both workers and the wider community are kept safe.
If you are looking to detect for Carbon Dioxide (CO2) view our range of gas detection monitoring equipment or contact us and one of our professionals will be able to help you.

